March 3, 2026 at 12:00 PM ET
Webinar Recap: Navigating the NIH 2026 Updates and Policy Changes
A walkthrough of recent NIH policy updates and submission changes.
Access Webinar Recording
Please fill out this form to access the recorded webinar.
Navigating federal funding is no longer a straightforward process of scoring well and securing awards, yet it remains a critical component of building a sustainable research enterprise. In a recent webinar Jennifer Troyer, a freelance science advisor and former NIH program director, shared a landmark framework for moving from reactive grant chasing to proactive funding strategy development in today's transformed landscape.
Here is a breakdown of her approach to adapting funding strategies during this period of confusion and chaos at the NIH.
The Current Reality
What Hasn't Changed
Despite all the upheaval, some fundamentals remain intact. The NIH mission continues unchanged, and the basic application process still follows the same path through CSR, peer review, and council approval. Most importantly, the dedicated staff who remain are still committed to supporting quality science and helping researchers succeed.
The Staffing Crisis
NIH has lost far more than the reported 20% staff reduction. The agency suffered a devastating 50% cut in contract workers, while all probationary employees were terminated overnight with no transition period. Some divisions, like Jennifer's former division at NHGRI, lost an staggering 2/3 of their staff.

Impact: Slower response times, communication gaps, limited guidance capacity.
Key Changes You Need to Know
The New Funding Reality
The most critical change is that fewer awards are going out despite similar overall budget levels. The era of predictable pay lines is over, meaning good scores no longer guarantee funding. The new multi-year funding mandate requires awards to be fully funded upfront, dramatically reducing the total number of grants that can be made. Perhaps most concerning, funding decisions now involve multiple political approval levels, adding uncertainty even to scientifically sound proposals.
Word Choice Matters
While there are technically no banned words, certain terms trigger additional layers of review that can delay or derail funding. DEI-related terms like diversity, equity, and inclusion now raise flags, as do health disparities language and any research related to vaccine hesitancy. Jennifer's strategic advice is clear: use alternative phrasing where possible, but be prepared to advocate strongly when certain terminology is essential to your scientific work.
Major Policy Changes
Several significant policy shifts are reshaping how researchers apply for funding. Foreign collaborations can no longer use traditional subawards and must instead receive separate awards using new PF5/UF5 activity codes. Individual PIs are now limited to 6 applications per year, regardless of funding mechanism. The agency has imposed new restrictions including a ban on AI-written grants and heavy limitations on gain-of-function research. All review processes have been centralized under CSR with a simplified scoring framework, potentially creating expertise gaps in specialized research areas.
Success Strategies
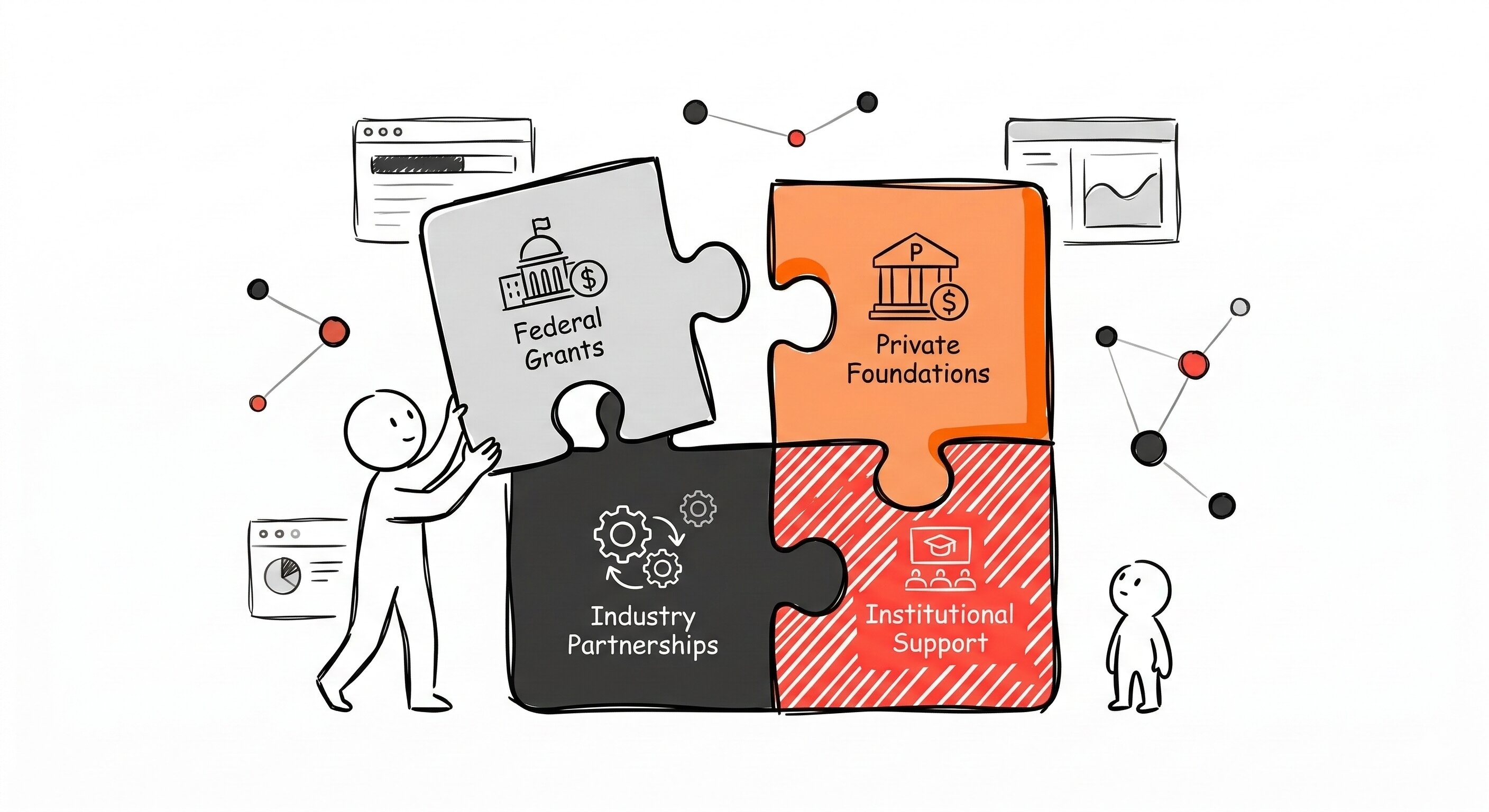
Diversify Your Funding
Jennifer's most emphatic recommendation is to stop relying solely on federal funding. Researchers must build stronger institutional partnerships, actively explore foundation and industry opportunities, and plan for significantly longer funding gaps. The days of predictable federal funding cycles are over, making diversification not just smart but essential for research sustainability.
Working with Reduced Staff
With severely reduced staffing levels, researchers must adapt their communication strategies. Ask only clear, specific questions and be patient with significantly longer response times. Always submit help desk tickets to create timestamps for your inquiries, and while persistence is important, maintain kindness in your follow-ups. Remember that the remaining staff are often handling multiple roles and facing unprecedented workloads.
Application Strategy
Successful applications now require more careful navigation than ever before. Start by talking directly with program directors about how your research fits within current funding priorities—this conversation is more critical now with reduced funding opportunities. Carefully review all language for potential flags that might trigger additional review, and where scientifically appropriate, align your work with stated administration priorities. Most importantly, prepare for significantly longer timelines and increased uncertainty throughout the entire funding process.
Access Jennifer's Presentation
To dive deeper into the insights discussed in the webinar, you can access Jennifer Troyer's full slide deck directly below.
Download Jennifer's Presentation: Adapting Proposal and Funding Strategies to Today’s NIH Landscape
Looking Forward
The NIH landscape continues evolving with more changes expected. Success in this new environment requires strategic adaptation to rapidly changing policies, genuinely diversified funding approaches, realistic expectation setting about what can be achieved, and an unwavering continued focus on scientific excellence.
Key Takeaway: Keep doing excellent science, adapt strategically, and build comprehensive funding portfolios beyond federal sources.
Jennifer Troyer
Science Advocate, Former Director, Division of Extramural Operations, NHGRI, NIH
Jennifer is a leading expert on NIH policy, funding strategies, and research administration compliance; dedicated to helping institutions navigate complex federal guidelines. Former NIH Program Director and Freelance Science Advisor.
Raphaël Bernier
Head of Growth, Atom Grants
Helping universities modernize research development with AI to reduce admin burden, increase faculty engagement, and improve proposal success.
LinkedIn: https://www.linkedin.com/in/raph-bernier/
Contact: raphael@atomgrants.com
Location: New York